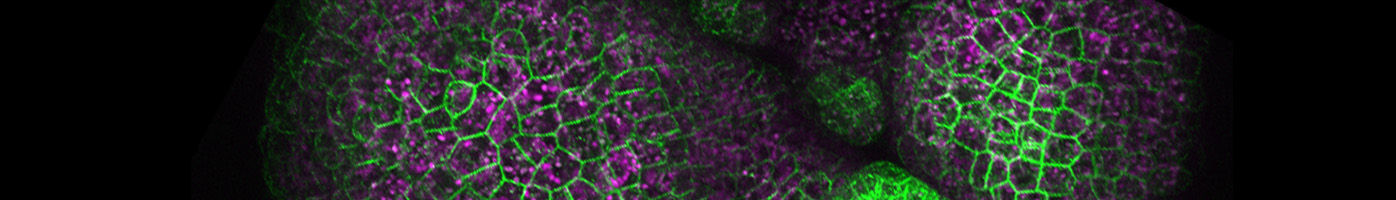

Underpinning the basic principles of self-organization using plant regeneration as a model
The extreme fantasies of regeneration paraded by many mythological and comic characters are encompassed in one single life form: the plant. Remarkable ability to repair the damaged body parts and to regenerate organs lost in injury is
prevalent across the kingdoms. While in animals this ability is restricted to specific lineages,
it is universal in plants. Plants not only regrow lost parts, but even regenerate an entire new
plant from a few pre-existing cells irrespective of their origin and developmental context. In
response to the damage caused by biotic and abiotic factors, cells in the vicinity of wound
must respond promptly and get reprogrammed to repair the damage. This is strikingly
different from animal kingdom, where specialized cells can be recruited to the wound site.
Plants are likely to deploy kingdom-specific mechanisms to deal with intrinsic differences
such as lack of cell migration, a key cellular process, extensively utilized in animal
regeneration. This makes plant regeneration an interesting model to underpin the basic
principles and dynamics of self-organization.
Our laboratory uses tissue-culture-mediated de novo shoot regeneration and innate ability of restoration of tissue or organ lost in injury as models to address fundamental questions related to acquisition of pluripotent state, reconstitution of stem cells, dynamics of cellular behaviour, assembly of regulatory interactions and gene regulatory network. Most of our inferences are based on live imaging of cytological events during regeneration. We use interdisciplinary approaches which combine molecular biological tools, genetics, genomics, bio-imaging and computational modelling to unearth the mechanisms of cellular reprogramming and developmental plasticity in plant.


Kalika Prasad, PhD
Professor (Group Leader)
Indian Institute of Science Education and Research, Pune.
Email: kalika.prasad@iiserpune.ac.in
After spending many years in Indian Institute of Science, Bangalore addressing how conserved regulators of inflorescence and flower development have acquired species-specific function in rice, I moved to Massey University, New Zealand and spent a year with a fatty friend gordita (regulator of ovule development). Soon after that I landed in the Netherlands (Utrech University) during cold dutch weather and had great fun admiring the beauty of nature and venturing into the regulation of lateral organ positioning in plants. Now here at IISER, our group explores the unparalleled ability of plants to regenerate specific cell types, tissues, organs and the whole organism. We have begun to unveil the mechanisms of this extra-ordinary developmental plasticity.
 Twitter
Twitter Google Scholar
Google Scholar Research Gate
Research Gate ORCID
ORCIDRecent Publications
Vacancies
Inquiries from prospective students, graduate trainees, postdocs and visitors are welcome.
Please email on kalika.prasad@iiserpune.ac.in with your detailed cv and an outline of your interests.